- Blog
- Keyshot 8 crack download
- Free ccleaner download
- Ghost of tsushima toyotama map
- Photoshop cc 2017 crack file amtlib-dll
- Bluestacks premium crack 2017
- Fonts that look like grateful dead download
- Full ccleaner free download
- Free bfme 2 cd key
- Personal budget spreadsheet excel
- Free daily expense excel tracker
- Super mystery dungeon pokemon -3ds rom decrypted
- Age of empire 3 full download-
- Free background zoom
- Weight tracker template free
- Free resume templates professional reddit
- Klondike solitaire freecell green felt
- Big fish mac games hidden object
- Minecraft xray 1-14-3 resource pack
- Google sheets meal plan ingredient
- Free credit card generator with real money
- Ps3 afterglow controller not working for pc
- Solve a quadratic equation using square roots
- Funeral homes in pleasant unity pa
- 1963 ford thunderbird convertible
- Solving quadratic equations by factoring examples
- 30 60 90 day sales training plan
- 3d us population density map
- Aplicacion pcclone ex lite
- Dewalt battery adapter ebay
- Resident evil 3 remake jill valentine mods
- Microsoft word fillable calendar template
- Duck life battle full game free download
- Macro meal planner for 1500 calories
- Minion butt crack
- Red giant serial keys
- Ea sports ufc 3 pc download key
- Gta 6 videos download
- Monthly office expense report template excel
- Population density map of major us cities
- Freecell games free online
- Dark age of camelot free shard
- -100000 mortgage payment calculator
- Origin 8 free download for windows 7
- Twilight forest mod download forge
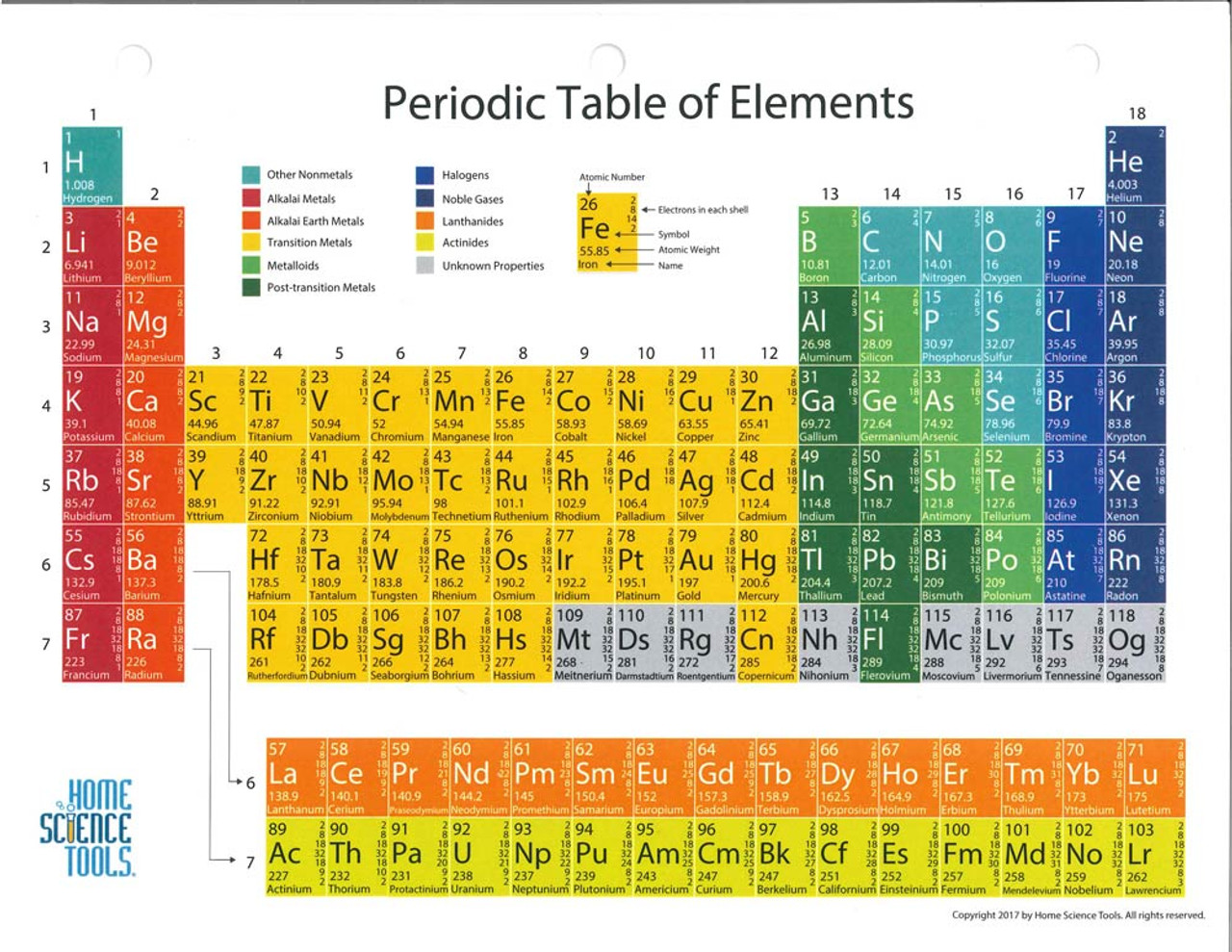
- Periodic table color coded fo group names
- Logitech g29 gaming software brake setting
/PeriodicTableallcolor-58b5c82c3df78cdcd8bbb80f.png)
Each element will be described in this order: atomic number name symbol whether it is a metal, metalloid, or nonmetal whether it is a solid, liquid, or gas and atomic mass. It is labeled “Symbol.” Below that is the number 1.008 which is labeled “Atomic Mass.” Below that is the word hydrogen which is labeled “name.” The color of the box indicates that it is a nonmetal. The number 1 is in its upper-left hand corner and is labeled “Atomic number.” The letter “H” is in the middle in red indicating that it is a gas. To the left of this box is an enlarged picture of the upper-left most box on the table. The 18 columns are labeled “Group” and the 7 rows are labeled “Period.” Below the table to the right is a box labeled “Color Code” with different colors for metals, metalloids, and nonmetals, as well as solids, liquids, and gases. doi: 10.1515/ract-2018-3082.\): Elements in the periodic table are organized according to their properties. "Evolution of the periodic table through the synthesis of new elements". Archived from the original on 30 January 2021. "The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update".
Quantum Mechanics: Non-Relativistic Theory. "The Positions of Lanthanum (Actinium) and Lutetium (Lawrencium) in the Periodic Table". Archived (PDF) from the original on 13 April 2021. "Provisional Report on Discussions on Group 3 of the Periodic Table" (PDF).
:max_bytes(150000):strip_icc()/PeriodicTablenamescolor-58b5d91e3df78cdcd8d03326.jpg)
Archived (PDF) from the original on 25 March 2012. "New Notations in the Periodic Table" (PDF). Later (1902), Mendeleev accepted the evidence for their existence, and they could be placed in a new "group 0", consistently and without breaking the periodic table principle. See also Group 3 element#Composition.ĭ Group 18, the noble gases, were not discovered at the time of Mendeleev's original table. Some sources follow a compromise that puts La–Lu and Ac–Lr as the f-block rows (despite that giving 15 f-block elements in each row, which contradicts quantum mechanics), leaving the heavier members of group 3 ambiguous. Arguments can still occasionally be encountered in the contemporary literature purporting to defend it, but most authors consider them logically inconsistent. General inorganic chemistry texts often put scandium (Sc), yttrium (Y), lanthanum (La), and actinium (Ac) in group 3, so that Ce–Lu and Th–Lr become the f-block between groups 3 and 4 this was based on incorrectly measured electron configurations from history, and Lev Landau and Evgeny Lifshitz already considered it incorrect in 1948. This makes the group somewhat exceptional.ī The 14 f-block groups (columns) do not have a group number.Ĭ The correct composition of group 3 is scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr), as shown here: this is endorsed by 19 IUPAC reports on the question. Hydrogen is not considered to be an alkali metal as it is not a metal, though it is more analogous to them than any other group. Elements of the group have one s-electron in the outer electron shell. A Group 1 is composed of hydrogen (H) and the alkali metals.
